Lesson 8- Exponents, Logarithms, and Optical Density
 There is no video for this section. Please refer to the notes to complete worked examples in this lesson.
There is no video for this section. Please refer to the notes to complete worked examples in this lesson.
In optics, we frequently work with numbers that span many orders of magnitude. Light intensity, transmission, energy, and absorption often change by factors of ten rather than by small linear amounts. Exponents and logarithms give us a compact, consistent way to represent and manipulate these large and small values.
This lesson builds the mathematical foundation needed to understand scientific notation, powers of ten, logarithmic relationships, and optical density. These tools are essential for working with lasers, filters, coatings, and radiometric quantities throughout optics and photonics.
Exponents and Powers of Ten
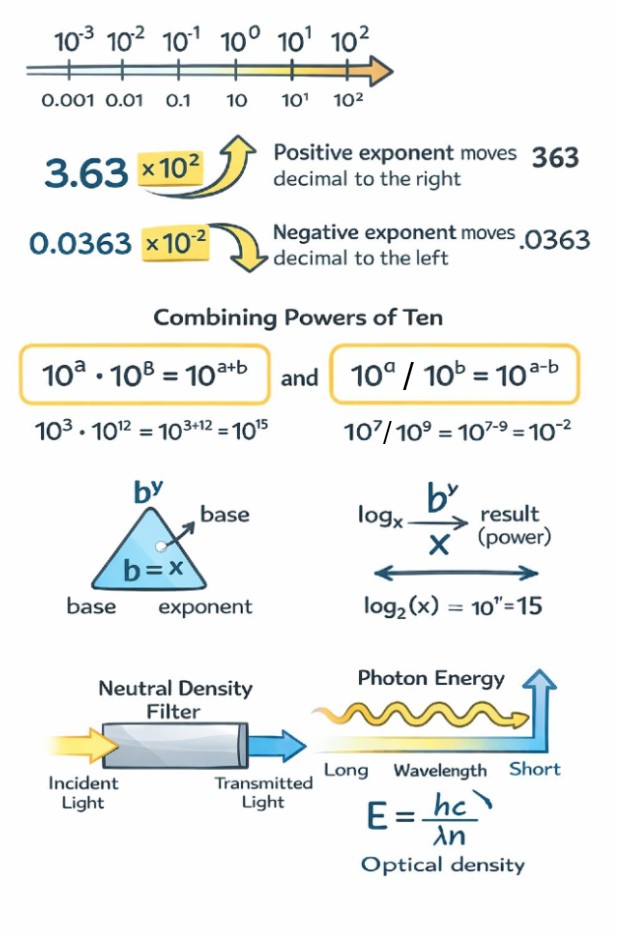
An exponent tells you how many times a number is multiplied by itself. When the base is 10, exponents provide a simple way to move the decimal point left or right. Positive exponents shift the decimal to the right, while negative exponents shift it to the left.
Several exponent rules appear repeatedly in optics calculations. Any nonzero number raised to the zero power equals one. A negative exponent represents a reciprocal. These rules allow large and small quantities to be expressed cleanly without writing long strings of zeros.
Worked Example: Powers of Ten
3.63 × 10² = 363
3.63 × 10⁻² = 0.0363
A positive exponent moves the decimal to the right.
A negative exponent moves the decimal to the left.
Multiplying, Dividing, and Raising Powers of Ten
When multiplying powers of ten with the same base, the exponents are added. When dividing, the exponents are subtracted. When a power of ten is raised to another power, the exponents are multiplied.
These rules allow complex expressions to be simplified quickly, which is especially important when working with scientific notation in multi-step optics problems.

(10⁷)(10⁹) = 10¹⁶
10⁻¹ · 10³ = 10² = 100
10⁰ = 1
 Logarithms as the Inverse of Exponents
Logarithms as the Inverse of Exponents
A logarithm answers the question: what exponent produces this number? Logarithms are the inverse operation of exponentiation. Writing an equation in logarithmic form makes it easier to solve for unknown exponents.
If x = bʸ, then logᵦ(x) = y. In optics, the most common logarithms use base 10 or base e.
Worked Example: Converting to Logarithmic Form
2⁴ = 16
log₂(16) = 4
2⁻² = 1/4
log₂(1/4) = −2
Common Logarithms and Natural Logarithms
When no base is written, log is assumed to mean base 10. These are called common logarithms and are widely used in optics, particularly in optical density calculations.
Natural logarithms use base e, where e is approximately 2.718. Natural logs are written as ln(x) and frequently appear in physics-based models and exponential decay processes.
Worked Example: Evaluating Logarithms
log(10⁴) = 4
ln(e²) = 2
ln(e⁰) = 0
Solving Exponential Equations Using Logarithms
Logarithms allow exponential equations to be rearranged so the unknown variable can be isolated. This technique is especially important when working with transmission, absorption, and decay equations in optics.
Worked Example: Solving for x
Given: y = 10³ˣ
Convert to logarithmic form:
log(y) = 3x
Solve for x:
x = log(y) / 3
Optical Density and Transmission
Optical density describes how strongly a filter or material attenuates light. It is related to transmission by a logarithmic relationship. As optical density increases, transmission decreases exponentially rather than linearly.
The relationship is:
T = 10⁻ᴼᴰ
where T is transmission and OD is optical density. A neutral density filter with an incoming beam and a visibly reduced transmitted beam.
Worked Example: Transmission from Optical Density
Given: OD = 0.7
T = 10⁻⁰·⁷
T ≈ 0.20
The filter transmits about 20% of the incident light.
Worked Example: Optical Density from Transmission
Given: T = 0.01
OD = −log₁₀(T)
OD = −log₁₀(0.01)
OD = 2
The optical density is 2.
Energy of Light and Wavelength
The energy of a photon depends on its wavelength. Shorter wavelengths correspond to higher energies. This relationship is fundamental in optics, spectroscopy, and laser safety.
The equation is:
E = hc / (λn)
where h is Planck’s constant, c is the speed of light, λ is wavelength, and n is the index of refraction.
Worked Example: Energy of Green Light
Given:
λ = 550 nm = 5.50 × 10⁻⁷ m
h = 6.626 × 10⁻³⁴ Js
c = 3.0 × 10⁸ m/s
n = 1
E = (6.626 × 10⁻³⁴)(3.0 × 10⁸) / (5.50 × 10⁻⁷)
E ≈ 3.61 × 10⁻¹⁹ J
The energy of the green light is approximately 3.6 × 10⁻¹⁹ joules.
Why These Concepts Matter
Exponents and logarithms are essential tools in optics, not abstract math. Optical density, transmission, and photon energy all rely on exponential relationships that describe how light behaves in real systems.
Understanding these concepts prepares you for work with lasers, optical coatings, filters, spectroscopy, and advanced optical analysis later in the course.